HOMEWORK help center ||HWH||open!
Re: HOMEWORK help center ||HWH||open!
Star Butterfly wrote:Catheren wrote:I HAVE A QUESTION!
TYPE OF QUESTION: Grammar
YOUR QUESTION: In this sentence "Considering the importance of clean water to the community the resounding question is how should we prevent saltwater intrusion in Monterey County?" should I leave it as is or should it read like this: "Considering the importance of clean water to the community the resounding question is 'How should we prevent saltwater intrusion in Monterey County?'"
I believe it is the second one
I think it depends on what country you're writing in. For American English, single quotes are rarely used and should be avoided. In British English, you can use single quotes to offset a word. Offsetting a whole phrase is uncommon, but you might get away with using single quotes to offset a phrase in British English.
In American English, the entire phrase, "how should we prevent saltwater intrusion in Monterey County," is meerly the object of the verb "is," so it does not need the single quotation marks.
If possible, I would re-word the sentence to avoid the issue. Below are some examples using different methods to offset that phrase. (additionally, I would recommend a comma after the word "community" because it marks the end of an introductory phrase, but I believe this comma is optional)
Considering the importance of clean water to the community, one resounding question remains: how should we prevent saltwater intrusion in Monterey County?
(In the above example, a colon is used to join two independent clauses when the second independent clause explains the first)
Considering the importance of clean water to the community, residents wonder how we should prevent saltwater intrusion in Monterey County.
(Here, I made the question a statement. Instead of stating the question, I state the subject (residents) and what they are doing (verb = wonder). The object of the verb is the question that you are asking.
Considering the importance of clean water to the community, residents want to prevent saltwater intrusion in Monterey County but do not know the best method for prevention.
(Here, I re-worded the question as if it were a statement of a problem. Although I am not sure if my statement is entirely accurate (you can fix it and make it accurate), this style of writing that avoids informal questions is generally expected in an introduction, which is where it sounds like this sentence came from.)
...............╭.....................................╮
...............Our deepest fear is not that we are inadequate.
...............Our deepest fear is that we are powerful
...............beyond measure. It is our light, not our darkness
...............that most frightens us. We ask ourselves, Who am I
...............to be brilliant, gorgeous, talented, fabulous?
...............Actually, who are we not to be?
...............~Marianne Williamson
...............╰.....................................╯
...............Our deepest fear is not that we are inadequate.
...............Our deepest fear is that we are powerful
...............beyond measure. It is our light, not our darkness
...............that most frightens us. We ask ourselves, Who am I
...............to be brilliant, gorgeous, talented, fabulous?
...............Actually, who are we not to be?
...............~Marianne Williamson
...............╰.....................................╯
-

username by me - Posts: 4796
- Joined: Sun Nov 18, 2012 12:54 pm
- My pets
- My items
- My wishlist
- My gallery
- My scenes
- My dressups
- Trade with me
Re: HOMEWORK help center ||HWH||open!
Thank you both!
-

Catheren - Posts: 14382
- Joined: Sun Jan 16, 2011 11:02 am
- My pets
- My items
- My wishlist
- My gallery
- My scenes
- My dressups
- Trade with me
Re: HOMEWORK help center ||HWH||open!
I HAVE A QUESTION!
TYPE OF QUESTION: precalc
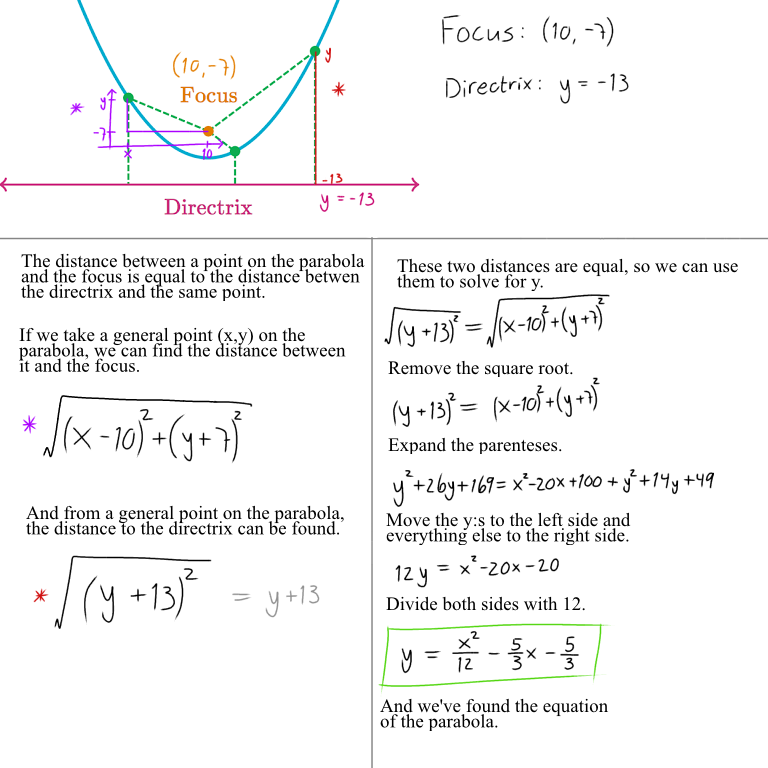
YOUR QUESTION: "find the standard form of the equation of the parabola."
focus at (10, -7), directrix y=-13
pls help we have a test on wednesday and i'm soo confused skk
TYPE OF QUESTION: precalc
YOUR QUESTION: "find the standard form of the equation of the parabola."
focus at (10, -7), directrix y=-13
pls help we have a test on wednesday and i'm soo confused skk
-

dovah - Posts: 9705
- Joined: Sun Nov 17, 2013 4:28 am
- My pets
- My items
- My wishlist
- My gallery
- My scenes
- My dressups
- Trade with me
Re: HOMEWORK help center ||HWH||open!
dovah wrote:I HAVE A QUESTION!
TYPE OF QUESTION: precalc
YOUR QUESTION: "find the standard form of the equation of the parabola."
focus at (10, -7), directrix y=-13
pls help we have a test on wednesday and i'm soo confused skk
I hope this helps some. If there is something that I haven't explained well, feel free to ask. This site could be worth a read too.
I made a small error at first, it has been corrected.
-

LostInTheEcho - Posts: 2910
- Joined: Wed Nov 05, 2014 9:43 am
- My pets
- My items
- My wishlist
- My gallery
- My scenes
- My dressups
- Trade with me
Re: HOMEWORK help center ||HWH||open!
I HAVE A QUESTION!
TYPE OF QUESTION: psychology
YOUR QUESTION: i'm currently taking pre-ib psychology and was looking for someone who is can answer my questions and help me understand difficult concepts!! hoping to ask frequent questions through pm and can definitely offer pets as payment <3 please pm me if you can help a psych student out
TYPE OF QUESTION: psychology
YOUR QUESTION: i'm currently taking pre-ib psychology and was looking for someone who is can answer my questions and help me understand difficult concepts!! hoping to ask frequent questions through pm and can definitely offer pets as payment <3 please pm me if you can help a psych student out



-

hannur - Posts: 1169
- Joined: Sat May 13, 2017 3:07 pm
- My pets
- My items
- My wishlist
- My gallery
- My scenes
- My dressups
- Trade with me
Re: HOMEWORK help center ||HWH||open!
I HAVE A QUESTION!
TYPE OF QUESTION:maths,
YOUR QUESTION:
Solve the following equation for 0≤ θ ≤ 360
2(sin θ ) ^ 2 - 3 sin θ + 1 = 0
thank you !
TYPE OF QUESTION:maths,
YOUR QUESTION:
Solve the following equation for 0≤ θ ≤ 360
2(sin θ ) ^ 2 - 3 sin θ + 1 = 0
thank you !
- ehearts
- Posts: 1
- Joined: Thu Aug 08, 2019 11:25 pm
- My pets
- My items
- My wishlist
- My gallery
- My scenes
- My dressups
- Trade with me
-

Waki - Posts: 5505
- Joined: Tue Feb 11, 2014 11:33 am
- My pets
- My items
- My wishlist
- My gallery
- My scenes
- My dressups
- Trade with me
Re: HOMEWORK help center ||HWH||open!
ehearts wrote:I HAVE A QUESTION!
TYPE OF QUESTION:maths,
YOUR QUESTION:
Solve the following equation for 0≤ θ ≤ 360
2(sin θ ) ^ 2 - 3 sin θ + 1 = 0
thank you !
This might be a bit late, but the idea is that you want to replace sin θ as x and solve it like a quadratic equation first. You can re-write the equation this way because sin θ is a function, so for any θ, you will get one and only one value of sin θ. Call that one value x.
If you re-write sin θ as x, you get the following:
2(x)^2 - 3x + 1 = 0
Now you can solve for x by factoring the equation:
(2x - 1)(x - 1) = 0
Now set each set of parentheses equal to zero and solve for x each time:
2x - 1 = 0 x - 1 = 0
2x = 1 x = 1
x = 1/2
(alternatively, you can use the quadratic equation to solve for x if factoring is not obvious to you)
Now that you have x = 1/2 and x = 1, replace sin θ with x and sole for θ:
sin θ = 1/2 sin θ = 1
θ = arcsin(1/2) θ = arcsin(1)
Now, just recall that arcsin(1/2) and arcsin(1) have fixed values with a domain of θ = [-90, 90].
If you use a calculator to solve this equation, you get the following:
arcsin(1/2) = 30 arcsin(1) = 90
However, you don't want values just between -90 and 90, you want values between 0 and 360. If you use the unit circle, you will notice that another angle will also give you arcsin(1/2), and that angle is 30 clockwise from the left horizontal, or 150 degrees (let me know if you need an explanation of why this is). There is no other angle that give you arcsin(1), also due to unit circle geometry.
Therefore, you have three angles between 0 and 360 that will solve this equation: 30, 90, and 150
...............╭.....................................╮
...............Our deepest fear is not that we are inadequate.
...............Our deepest fear is that we are powerful
...............beyond measure. It is our light, not our darkness
...............that most frightens us. We ask ourselves, Who am I
...............to be brilliant, gorgeous, talented, fabulous?
...............Actually, who are we not to be?
...............~Marianne Williamson
...............╰.....................................╯
...............Our deepest fear is not that we are inadequate.
...............Our deepest fear is that we are powerful
...............beyond measure. It is our light, not our darkness
...............that most frightens us. We ask ourselves, Who am I
...............to be brilliant, gorgeous, talented, fabulous?
...............Actually, who are we not to be?
...............~Marianne Williamson
...............╰.....................................╯
-

username by me - Posts: 4796
- Joined: Sun Nov 18, 2012 12:54 pm
- My pets
- My items
- My wishlist
- My gallery
- My scenes
- My dressups
- Trade with me
Re: HOMEWORK help center ||HWH||open!
I HAVE A QUESTION!
TYPE OF QUESTION:chemistry
YOUR QUESTION:okay so this is kinda a lot but any help on any question would be VERY appreciated! im completely clueless. i’m taking AP biology, i’ve never taken chemistry in my life so i’m completely clueless. this is all of the “essential” chemistry for ap bio.
-

questions about water, properties of water. i’m not sure if the ones i could answer are correct. i can’t find anything online specific enough to answer these questions fully.
-

molarity concentration, looks like math maybe?
-

more molarity concentration and something about pH
-

isomers and functional group questions. i have no clue what these things mean.
-
if you can only help with one question i would really appreciate it. thank you for taking the time to read this all.
TYPE OF QUESTION:chemistry
YOUR QUESTION:okay so this is kinda a lot but any help on any question would be VERY appreciated! im completely clueless. i’m taking AP biology, i’ve never taken chemistry in my life so i’m completely clueless. this is all of the “essential” chemistry for ap bio.
-

questions about water, properties of water. i’m not sure if the ones i could answer are correct. i can’t find anything online specific enough to answer these questions fully.
-

molarity concentration, looks like math maybe?
-

more molarity concentration and something about pH
-

isomers and functional group questions. i have no clue what these things mean.
-
if you can only help with one question i would really appreciate it. thank you for taking the time to read this all.
bellamare
adult - it/she - pansexual - canine therian
part time student, vet tech and furry artist
links
lf art
art shop
ocs ufs/uft
-

bellamare - Posts: 7742
- Joined: Wed Jul 01, 2015 3:24 am
- My pets
- My items
- My wishlist
- My gallery
- My scenes
- My dressups
- Trade with me
Re: HOMEWORK help center ||HWH||open!
long shoe wrote:I HAVE A QUESTION!
TYPE OF QUESTION:chemistry
YOUR QUESTION:okay so this is kinda a lot but any help on any question would be VERY appreciated! im completely clueless. i’m a junior taking AP biology, i’ve never taken chemistry in my life so i’m completely clueless. this is all of the “essential” chemistry for ap bio.
- Ooh, stoichiometry! I'd have to review the biochem and functional groups, that's not my forte, but I can explain parts of this.
Earlier on the page you said that molarity is moles of solute over liters (of solution). Great! Write that on your paper a few more times, if it helps, since that's the key for the next few questions.
19.C) 1. 10.0g of sulphuric acid + 0.1L of water, find the molarity.
Here, you're given grams of H2SO4. Since that's the solute (the compound being dissolved), and you need to know the quantity of moles of the solute to be able to find molarity, find the equivalent of 10g in moles.
To do this, divide the mass (10g) by the molar mass of H2SO4. (Why? Check with a unit conversion; you're trying to find moles. (g)/(g/mol) = (g)(mol/g), grams cancel out = moles.)
Molar mass of H2SO4 = the combined masses of all its components = (2 x molar mass of hydrogen) + (1 x molar mass of sulphur) + (4 x molar mass of oxygen).
Google tells me it's 98.08g/mol, but make sure you know where on the periodic table you can find these masses during your tests, and check that you are able to calculate the correct final mass several times.
Okay, 10g/98.08g/mol = 0.102mol of H2SO4.
Molarity = moles of solute / litres of solution, and you've got both now.
0.102mol H2SO4 / 0.1L H2O = 1.02M.
(Note, capital M is the symbol for molarity - I think that was another question for you.)
Does this make sense? The other questions under 19C are either very similar, or the same with the reverse process.
Try out another one, feel free to let me know if you have questions. c:
--
15.
Try this link - https://science.howstuffworks.com/envir ... s/h2o7.htm, it has pretty much all the terms you're looking for.
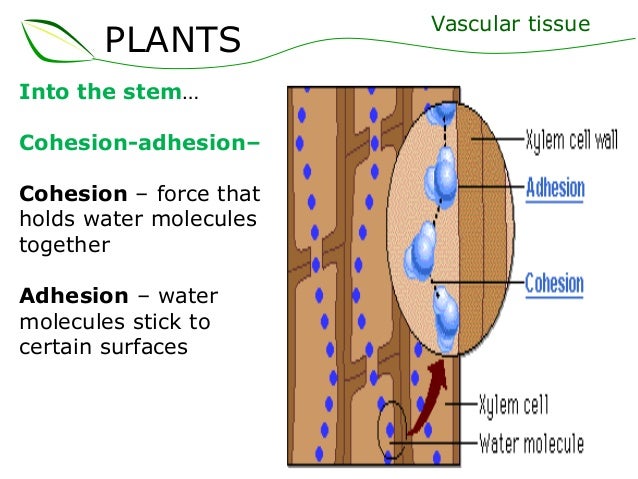
Cohesion and adhesion - if you're in bio, the example you'll see at some point is water travelling up plant xylem. Cohesion is the water particles sticking to each other, adhesion is the particles sticking to other particles. Therefore, in the plant stem, the water adheres to the inside of the stem, and also coheres to itself. (As an aside, this is the first time I have ever used the verb 'to cohere' - I had to search up whether it exists, but it does!)
--
16. Water is unique in that the solid state is less dense than the liquid state. This happens because ice is organized into a lattice while water is relatively mobile:

Try to search up the why if you're interested in looking at this further; I suggest this link, https://chemistry.stackexchange.com/que ... than-water, but it may be too technical.
--
Please do more research into these! It's almost midnight here and I'm just giving you a place to start, since you said you couldn't find anything specific online.
17b. "What is it about water molecules and the ions in salt that might make water able to dissolve salt?
The positive and negative polar ends of a water molecule are attracted to the negative chloride ions and positive sodium ions in the salt." from https://www.middleschoolchemistry.com/l ... r5/lesson3.
The polar ends pull NaCl (and other ionic compounds) apart. For larger molecules like sucrose, try here: https://chemed.chem.purdue.edu/genchem/ ... oluble.php
c. https://www.usgs.gov/special-topic/wate ... er_objects
d. Basically, sweat is primarily water (with some salts and other components), and water has a high specific heat capacity (requires a lot of energy/heat to heat up). So, your body exudes this water onto the skin, heats it up (therefore using up body heat), the water in the sweat evaporates and takes the heat it used to do so with it.
e, f. http://www.appstate.edu/~goodmanjm/rcoe ... waterdrops
g. Same concept as water moving up a plant stem. https://www.usgs.gov/special-topic/wate ... er_objects
Last edited by CᴀɴᴅʏNᴜᴛᴍᴇɢ on Mon Dec 23, 2019 8:38 pm, edited 1 time in total.
-

CᴀɴᴅʏNᴜᴛᴍᴇɢ - Posts: 802
- Joined: Mon Nov 12, 2012 1:10 pm
- My pets
- My items
- My wishlist
- My gallery
- My scenes
- My dressups
- Trade with me
Who is online
Users browsing this forum: No registered users and 1 guest



















